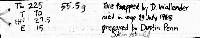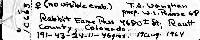|
|
|
|
Family: Mustelidae
Ermine, more... (fr: hermine)
|
Reese Galvin Mustela erminea in Arizona: Ecological Physiology and Similar Species: Ecological Physiology Mustela erminea, commonly called the stoat, is a member of the Mustelidae family, and is one of the most widely distributed mustelids across the globe. It is a mammal that lives primarily in cold and wet environments, but has a very wide range of habitats. Stoats live for roughly 4 years in the wild (LaPoint et al. 2017). They have been known to swim to escape predators (Veale 2013) and often reside in the burrows of their recently killed rodent prey rather than building or digging their own shelters. It is generally thought that they get their water from their diet of small rodents, although virtually no observational research has been performed on the drinking habits of Mustela erminea. Their body size may have a positive correlation with temperature and they may not follow Bergmann’s rule (Erlinge 1987), which states that in general, larger species should live in colder environments and smaller species should live in warmer climates. Instead, the stoat’s body size varies greatly regardless of its habitat, which may be due to its feeding habits or adaptation to climatic conditions (Erlinge 1987). While no studies have yet determined the minimum and maximum survivable temperatures for M. erminea, its inability to enter torpor and its active winter behavior (LaPoint et al. 2017) which defy Bergmann’s rule indicate that its range may be similar to its smaller cousin Mustela nivalis’ range of -20C to 20C (Zub et al. 2013). In winter, the Stoat’s coat whitens to match its surroundings in order to protect against predation. This is achieved via temperature sensing in the pituitary gland inhibiting synthesis of the melanocyte-stimulating hormone preventing brown hair from growing (King and Moody 1982). For this reason, farther-north stoats tend to undergo color change sooner than southern stoats, and in fact, some southern stoats do not actually complete the color change before spring (King and Moody 1982). Daylength was first suggested to play a role in the molting process by Bisonnette and Bailey in 1944 and was further supported by research which found that anagen phases coincided with changes in daylength, with northernmost stoats beginning their molting phases sooner than their southern counterparts (King and Moody 1982). Because stoats are unable to hibernate or enter torpor, they instead reduce body mass to lower energy expenditure. In fact, their behavior changes very little in the winter and they are still considerably active in hunting and foraging efforts in comparison to larger mammals (LaPoint et al. 2017). Similar Species The closest related species to Mustela erminea is M. nivalis, the least weasel. Though the stoat is much larger than the weasel, they generally coexist and in fact share an almost identical habitat range throughout the world (Erlinge and Sandell 1988). Neogale frenata, the long-tailed weasel, is another species similar to M. erminea. Neogale frenata is larger and once again is just as widely distributed as the other two, and in fact they all three live in relative coexistence with each other. The size differences between them ensure that the prey they share are pretty evenly distributed among populations, as the smaller M. nivalis can burrow deeper into the smaller tunnels of the rodent prey, and N. frenata consumes large rodents that the other two species could not (St. Pierre et al. 2006). Neogale frenata is generally considered to be the only mustelid that resides in Arizona, primarily inhabiting the higher elevated areas of the state. The one recorded sighting of Mustela erminea in Arizona occurred in 1989, near Jacob Lake in Coconino County (Berna 1991). M. nivalis has not yet been identified in Arizona. These species generally tend to reside in the more northern areas of North America, though it is possible that populations may exist in Eastern Arizona and Northern Arizona, but more research is required to make a definitive statement as to whether or not M. erminea and M. nivalis inhabit the area. If one were to encounter a mustelid in the field, the most identifiable and distinguishing features that would separate the specimens from each other would be the body size and tail lengths relative to body size. Mustela nivalis is the smallest of the three, with a maximum tail length of up to 30% of its total body length, which is typically between 130 and 260mm (Heptner and Sludskii 2002). M. erminea’s body is generally sized in between M. nivalis and N. frenata, with a tail approximately 30% of its body length of 175 and 325mm (Harris 2008) with some upward variation (Heptner and Sludskii 2002). Neogale frenata has the largest body at 300-350mm and its tail length reaches up to 70% of its total body length (Sheffield and Thomas 1997). REFERENCES: Berna, H. J. 1991. First record of the ermine (Mustela erminea) in Arizona. The Southwestern Naturalist 36:245. Erlinge, S. 1987. Why do European stoats Mustela erminea not follow Bergmann's rule? Ecography 10:33–39. Erlinge, S. and M. Sandell. 1988. Coexistence of stoat, Mustela erminea, and weasel, M. nivalis: Social dominance, scent communication, and Reciprocal Distribution. Oikos 53:242. Harris, S. 2008. Mammals of the British Isles: Handbook. Mammal Review 38:327–328. Heptner, V. G. and Sludskii, A. A. 2002. Mammals of the Soviet Union. Vol. II, part 1b, Carnivores (Mustelidae and Procyonidae). Smithsonian Institution Libraries and National Science Foundation. King, C. M., & Moody, J. E. 1982. The biology of the stoat (Mustela erminea) in the National Parks of New Zealand. New Zealand Journal of Zoology, 9(1), 103-118. LaPoint, S., L. Keicher, M. Wikelski, K. Zub and D. K. Dechmann. 2017. Growth overshoot and seasonal size changes in the skulls of two weasel species. Royal Society Open Science 4:160947. Sheffield, S., H. Thomas. 1997. Mustela frenata. (11 November 2022). St. Pierre, C., J.-P. Ouellet and M. Crête. 2006. Do competitive intraguild interactions affect space and habitat use by small carnivores in a forested landscape? Ecography 29:487–496. Veale, A. J. 2013. Observations of stoats swimming. New Zealand Journal of Zoology 40:166–169. Zub, Karol, et al. “Male Weasels Decrease Activity and Energy Expenditure in Response to High Ambient Temperatures.” PloS ONE 8(8):e72646.
Morgan Wagner Mustela erminea in Arizona: Distribution and Human Interactions: Distribution and habitat (AZ and global) The ermine, Mustela erminea is a small carnivorous mammal that belongs to the family Mustelidae (King and Powell, 2007). Mustela erminea has a wide range of distribution globally (King and Powell, 2007). The ermine can be found in the circumboreal range throughout the holarctic (King, 1983). The modern species of ermine first appeared around 0.6 million years ago in Eurasia (King and Powell, 2007). Then about half a million years ago Mustela erminea entered North America through the Bering Land Bridge (King and Powell, 2007). Mustela erminea is now native to Europe, Asia, and North America and has been introduced by humans to New Zealand (King, 1983). Mustela erminea inhabit a variety of habitats and can be found in forests, grasslands and tundras (King, 1983). However, Mustela erminea favor high elevation riparian habitats (Frey and Calkins, 2010). Because Mustela erminea is well adapted to the cold, ermine can be found in many regions with prolonged winters (King and Powell, 2007). Climate change during the Pleistocene period resulted in the formation of ice glaciers in North America, forcing entire ecosystems to move southward (King and Powell, 2007). This caused Mustela erminea to migrate into various mountainous regions throughout the southwestern United States (King and Powell, 2007). However, Mustela erminea is considered to be extremely rare in New Mexico and surrounding regions (Frey and Calkins, 2010). The first record of Mustela erminea in Arizona was found by Howard Berna at Fracas Lake, Arizona in 1991 (Berna, 1991). This specimen is now in the Arizona State University mammal collection (Berna, 1991). Interactions with humans/conservation concerns According to the IUCN, Mustela erminea is an abundant species with a wide circumboreal distribution that faces no significant threats. This is why the conservation status for Mustela erminea has been deemed of least concern (IUCN, 2016). However, in the southwestern United States Mustela erminea face many threats, including but not limited to habitat fragmentation, isolated populations, restricted distribution, low abundance, sensitivity to habitat alterations, the loss of the beaver, and climate warming (Frey and Calkins, 2010). Ermine are great predators and Mustela erminea is used by humans to exterminate pests such as rodents and rabbits (King and Moody, 1982). For this reason, humans have introduced Mustela erminea to New Zealand (King and Moody, 1982). The introduction of this species to New Zealand has had a negative impact on the native New Zealand wildlife (King and Moody, 1982). This is because Mustela erminea is not native to this region and when pest numbers are low, Mustela erminea choose to prey on native bird species (King and Moody, 1982). This resulted in the extinction or the endangerment of many bird species that are native to New Zealand (King and Moody, 1982). In order to monitor ermine populations, humans began trapping Mustela erminea. Captured Mustela erminea are essential to advancements in pest control (McDonald, 2002). Captive populations of Mustela erminea are used to test pest bait, lures, and traps. Utilizing captured Mustela erminea can improve rodent control technology and can provide a better landscape for human agriculture (McDonald, 2002). Mustela erminea is a disease vector for the Covid-19 virus and this poses a potential threat to humans (Ren et al., 2021). Viruses in mammalian hosts are largely misunderstood by the scientific community. Over the last few years the animal-to-human transmission of the Covid-19 virus has spiked interest in further understanding viruses in mammals. In April, 2020 Covid-19 patients transferred SARS-CoV-2 to mustelids in the Netherlands (Ren et al., 2021). Later, the infected mustelids transferred a new variant of Covid-19 (miSARS-CoV-2) back to humans (Ren et al., 2021). Ren and his colleagues then conducted a study and demonstrated that Y453F is an adaptive mutation that increased virus interaction in Mustela erminea and other mustelids ACE2 receptors, without compromising the viability of the human ACE2 receptors (Ren et al., 2021). This a potential fitness advantage for Mustela erminea and other mustelids and can increase the risk of animal-to-human SARS-CoV-2 transmission (Ren et al., 2021). REFERENCES: Berna, H. J. 1991. First record of the ermine (Mustela erminea) in Arizona. The Southwestern Naturalist 36:245. Frey, J. K., & Calkins, M. T. 2010. Status of the ermine Mustela erminea at its southern range limits, New Mexico. Final report to New Mexico Department of Game and Fish, Santa Fe, NM, USA:3-37. King, C. M. 1983. Mustela erminea. Mammalian Species:1-8. King, C. M., & Moody, J. E. 1982. The biology of the stoat (Mustela erminea) in the National Parks of New Zealand. New Zealand Journal of Zoology 9(1):103-118. King, C. M. and R. A. Powell. 2007. The Natural History of Weasels and Stoats. Ecology, Behavior, and Management. Oxford University Press, USA:13-23. McDonald, R. A., Webbon, C., & Harris, S. 2000. The diet of stoats (Mustela erminea) and weasels (Mustela nivalis) in Great Britain. Journal of Zoology: 252(3):363-371. Reid, F., Helgen, K. & Kranz, A. 2016. Mustela erminea. The IUCN Red List of Threatened Species. Ren, W. et al. 2021. Mutation Y453F in the spike protein of SARS-COV-2 enhances interaction with the mink ACE2 receptor for host adaptation. PLOS Pathogens:1-17. |
|
|
|